Loading...
Whatever your sterilisation needs Andersen have a solution either in house or through sterilisation services in Essex. Andersen’s continual research and product development ensures that our state-of-the-art systems deliver high levels of sterility assurance through a safe, efficient and highly reliable process. Andersen Products is now the only company offering Ethylene Oxide sterilisation cycle times as low as 3 hours. Aeration in the cabinet reduces residual EtO levels, so even a flexible endoscope can be in use again in 3½ hours.
Andersen Products offer solutions adapted to the sterilisation needs of veterinarians, hospital, laboratories and device manufacturers.
The Andersen Ethylene Oxide (EtO) sterilisation system delivers the gas in a small glass ampoule and holds the load in a flexible chamber within a cabinet that is below room pressure. This elegant design allows EtO sterilisation to be: accessible, safe, effective and efficient.
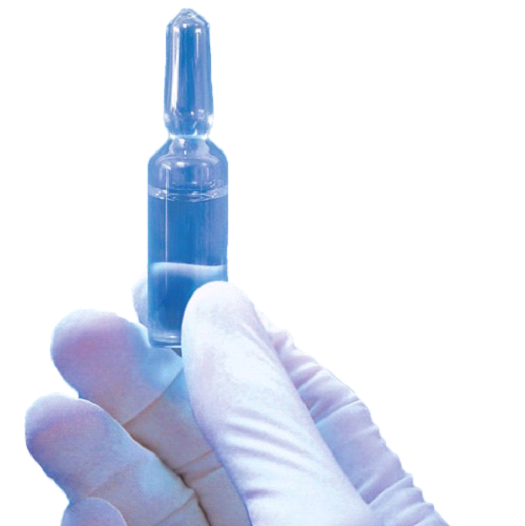
In the Andersen system the EtO is supplied in a glass ampoule. In the Anprolene refill kits the glass ampoule is held within a break shield. In the EOGas refill kits the ampoule is held within a plastic cartridge.
The ampoule avoids the use of compressed gas cylinders and the health and safety issues, and cost, associated with that.
Delivers gas to the heart of the load, achieving the highest level of sterilisation for any given amount of gas.
The gas is released by breaking the neck of the ampoule. The EtO boils, giving a very distinctive bubbling and cooling feeling as it does so. This provides very clear, positive feedback that the ampoule has been properly activated.

The Sterilisation chamber in the Andersen system is not the cabinet, but rather it is the bag. The packaged items, ready to be sterilised, are placed inside this sterilisation bag.
The cabinet holds the space around the sterilisation bag below room pressure. This ensures that any leak is from the room into the cabinet. Any EtO released through the bag is taken to the outside through the exhaust tube at the back of the cabinet.
The bag volumes vary in size up to 60 l, giving great flexibility in the volume, size and shape of items that can be sterilised.

The Anprolene and Series 4 sterilisers use a purge probe. The sterilisation bag is sealed around the bobbin on the purge probe.
At the start of the cycle the purge probe is used to draw any excess air out of the sterilisation bag. This achieves a high level of sterility using a very small amount of EtO.
At the end of the cycle the purge probe is used to “wash” clean air over the load, to reduce EtO residual levels. Air enters through the bobbin and passes down the probe, before being drawn back around the packages in the load and then out of the exhaust tube.

A unique chemical integrator has been developed by Andersen Products, with specific versions for use at room temperature and at 50 °C. This integrates the effects of gas concentration, temperature and time. So a colour change from orange to blue proceeds up the tube and signals a successful cycle when it passes the blue triangle on the pink backing plate.
This allows the operator to measure, with a ruler, how far the blue colour has gone in relation to the blue triangle. Within all other integrators you can only make some subjective assessment as to the colour change achieved, compared to that indicating a successful cycle. So, an operator who runs a load with highly gas absorbing items can see that the dosimeter has only just passed the mark, and adjust their future loads accordingly.
The Andersen system makes EtO technology accessible on a small scale, either for in house sterilisation or for small lot contract sterilisation. The elegant design means cabinets are available for under £ 4,000, with the most expensive cabinet selling for under £ 30,000. The consumables for all of the cabinets come at around £ 20 per cycle. The size of the Anprolene and Series 4 cabinets means they fit on a standard workbench. The largest, free-standing, cabinet only has a 0.7 m2 footprint. The size and weight of the Andersen cabinets greatly facilitates their installation. Installation involves installing the exhaust tubing, which amounts to making one hole to the outside, and plugging into a wall socket for power. There are very, very few restrictions on EtO emissions to atmosphere around the world.
The delivery of the EtO to the heart of the load in means that Andersen sterilisers use very small quantities of EtO. The largest ampoule holds just 17.6 g of EtO, which makes the whole system inherently much safer than any other EtO steriliser. Holding the sterilisation bag within a chamber that is below room pressure, effectively places the Andersen sterilisation chamber within its own safety room. Coupled with the many fail safe features and warnings built into the Andersen cabinets it is difficult to see how an operator would ever become exposed to EtO. Aeration within the cabinet ensures that the operators are not exposed to residual EtO. Whether the devices are immediately suitable for use will depend on what they are made of and what they are to be used for. In the Andersen system even the most difficult devices may be suitable for use on a patient after 24 hours at 20 °C or as little as 1 hour at 50 °C.
The EtO has long been recognised as a very effective sterilant. It remains the most common form of sterilisation worldwide for single use devices. The Andersen system makes this tried and trusted technology available on a small scale. The Sterility Assurance Level (SAL) of 10-6 which is the global standard for sterilisation, as opposed to disinfection, is available through the Andersen system to small operations and for small loads. In addition, due to its ability to pass through plastics, EtO is uniquely suitable for long tubes and complicated shapes. Where other technologies require adaptors and special cycles for difficult shapes, the Andersen system allows users to benefit from a fundamental property of EtO.
The use of the Andersen micro-dose system through the delivery of gas in a glass ampoule allows the Andersen system to be much more efficient in its use of EtO than any other system. This has benefits in terms of safety for the patient, the operator and the environment. If you start with a very small amount of EtO it is inevitably easier and quicker to ensure that neither the patient nor the operator are exposed to EtO. EtO will degrade in the environment, reacting with any organic matter or with water. That degradation happens more quickly and more safely if you start by using a much smaller quantity of EtO in the first place.